 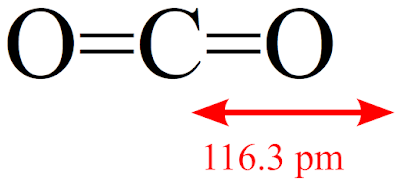
So, if you do these calculation, you will indeed found out that (at least in their ground electronic states) CO2 and H2O molecules have the equilibrium geometry mentioned in books.īut even for rigid (under specified conditions) molecules, one should remember, that even at the absolute zero of temperature nuclei are constantly vibrating near the equilibrium. We should be careful, since in general (as we have already said) we are not guaranteed that there exist only one minimum, but for small molecules such as CO2 and H2O that is not an issue. We can do what is known as the geometry optimization: starting from some initial geometry we try to minimise its electronic energy (including nuclear-nuclear repulsion energy) by varying arrangement of nuclei. 
And we can calculate equilibrium geometries for, say, ground electronic states, in the Born-Oppenheimer approximation. And some relatively small external influences can significantly change the molecular geometry.īut how small is small? And how big should be the barrier between two minima so that we say they are well separated? All this depends on the problem at hands and the corresponding physical conditions.įor instance, if we are talking about molecules in a gas phase under usual temperatures (300 K), then, say, ethane is non-rigid molecule, since its conformations are separated by just a few kJ/mol (comparable to energies of thermal motion), and thus, the molecular geometry of ethane constantly changes.īut, aside from this, for rigid (under usual conditions) molecules, like CO2 and H2O, we can meaningfully speak about their equilibrium geometries. Now, clearly, to meaningfully speak about equilibrium geometry for a molecule in a particular electronic state it is required that different minima on the corresponding PES are well separated, or, in other words, that there exist one distinct minimum on the potential energy surface.Īnd this is again not always the case: there exist some non-rigid molecules, for which minima are not well separated, or, in other words, which have few equilibrium geometries. Note, however, that even when PES for a particular electronic state of interest is well separated from PESes corresponding to other electronic states, there might exist more than one minimum on the same PES. This approximation breaks out when two PESes come close to each other or even intersect, but it is generally accurate at least for molecules in their ground electronic state (i.e. In this approximation the state (or, speaking classically, the motion) of electrons is treated independently from that of nuclei, and each electronic state of a molecule there exist the corresponding PES. Secondly, potential energy surface (PES) is a mathematical abstraction that appears only in approximate treatments of molecular systems within Born–Oppenheimer approximation. 
Each non-bonding pair is distributed over both oxygen atoms at once in molecular orbital theory, while in Lewis theory each lone pair is isolated to one atom or to localized bonds attached to that atom.Disclaimer: I will try my best to make this rather long story short.įirst of all, we must say that we are talking about the so-called equilibrium geometry - molecular geometry that corresponds to the true minimum on the potential energy surface, a surface which describes the energy of a molecule as a function of nuclear coordinates. Still, notice that each orbital is spread across both oxygen atoms at once, and again we see that each non-bonding electron pair in the HOMO is very different in molecular orbital theory compared to Lewis theory. \( \newcommand\) molecular orbitals, which are truly non-bonding and mostly oxygen in character. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
